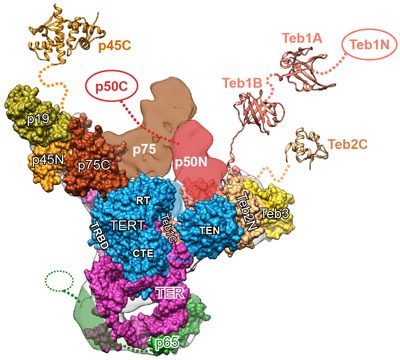
Like the features of a cat in a dark alley, those of an important enzyme called telomerase have been elusive. Using a combination of imaging techniques, a research team led by Juli Feigon ![]() of the University of California, Los Angeles, has now captured the clearest view ever of the enzyme.
of the University of California, Los Angeles, has now captured the clearest view ever of the enzyme.
Telomerase maintains the DNA at the ends of our chromosomes, known as telomeres, which act like the plastic tips on the ends of shoelaces. In the absence of telomerase activity, telomeres get shorter each time our cells divide. Eventually, the telomeres become so short that the cells stop dividing or die. On the other hand, cells with abnormally high levels of telomerase activity can constantly rebuild their protective chromosomal caps. Telomerase is particularly active within cancer cells.
Discovering how to turn telomerase off in cancer cells could lead to the development of new treatments that help prevent the diseased cells from multiplying. But doing this will require deep knowledge of telomerase’s structure.
“Many details we could only guess at before, we can now see unambiguously,” says Feigon. “If telomerase were a cat, before we could see its general outline … now we can see the details of the limbs; we can see the eyes, the whiskers, the tail and the toes.”
Scientists had thought telomerase contains seven proteins and RNA. In addition to showing where the RNA interacts with proteins as well as the shapes of some of those proteins, the researchers discovered two previously unknown proteins that increase the enzyme’s activity. The team’s findings were published last month in Science and are described in a UCLA news release ![]() .
.
“The day we realized what these new proteins are was amazing, knowing we were the first people in the world who knew about them!” Feigon recalls. “Days like that are what scientific discovery is all about, and it’s exhilarating!”
The scientists used as a model organism for their research a tiny, single-celled microorganism called Tetrahymena thermophila that is commonly found in fresh water ponds. Tetrahymena telomerase is remarkably analogous to human telomerase, the researchers have learned. Telomerase was discovered in this microorganism by Elizabeth Blackburn, who shared the 2009 Nobel Prize in medicine or physiology for her research on telomeres and telomerase.
The new findings represent more than eight combined years of research and brought together experts in structural biology, biochemistry and biophysics, who used several cutting-edge scientific techniques to reveal the enzyme’s mysteries.
Feigon and her colleagues are working to fill in even more details of the telomerase puzzle. “We can see the whiskers,” she says, “but cannot yet count the individual whiskers; we can see the toes, but not the nails.”


What the below report claims is that, “23andme,” have identified genes that I have, that are associated with longer or shorter telomere length. They list 5 gene variants that I have and say how much each could be expected to add to or subtract from my telomere length. We can do the math below to see how they may add up:
– 7.82 – 3.91 – 3.14 + 0 + 3.14 = – 11.73
So they are saying that based on the effect of these 5 genes my telomeres would be expected to be shorter as if I were 11.73 years older than my actual age.