Cells are the basic unit of life—and the focus of much scientific study and classroom learning. Here are just a few of their fascinating facets.
3.8 billion
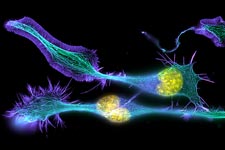
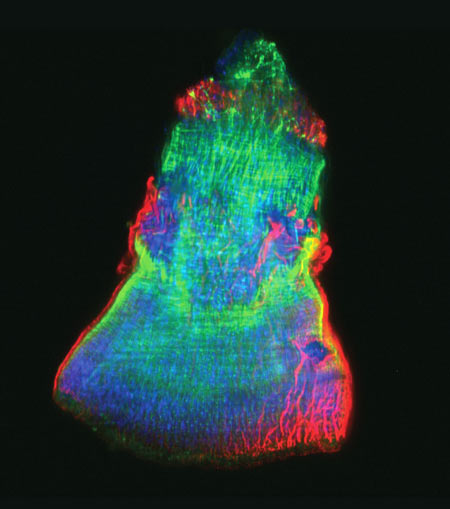
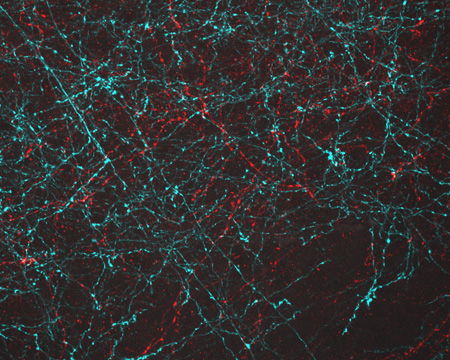
Developing nerve cells, with the nuclei shown in yellow. Credit: Torsten Wittmann, University of California, San Francisco.
That’s how many years ago scientists believe the first known cells originated on Earth. These were prokaryotes, single-celled organisms that do not have a nucleus or other internal structures called organelles. Bacteria are prokaryotes, while human cells are eukaryotes.
0.001 to 0.003
This is the diameter in centimeters of most animal cells, making them invisible to the naked eye. There are some exceptions, such as nerve cells that can stretch from our hips to our toes, sending electrical signals throughout the body.
1665
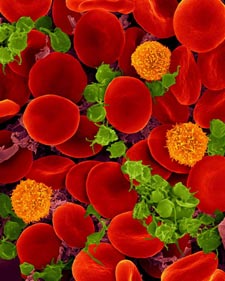
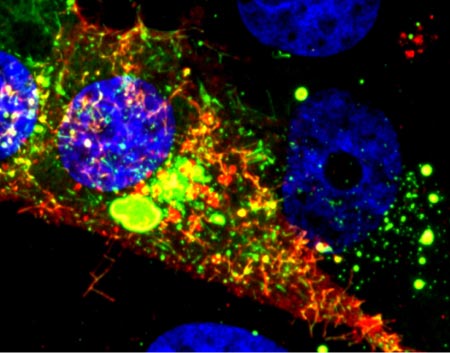
Oxygen-transporting red blood cells. Credit: Dennis Kunkel, Dennis Kunkel Microscopy, Inc.
In that year, British scientist Robert Hooke coined the term cell to describe the porous, grid-like structure he saw when viewing a thin slice of cork under a microscope. Today, scientists study cells using a variety of high-tech imaging equipment as well as rainbow-colored dyes and a green fluorescent protein derived from jellyfish.
200
That’s how many different types of cells are in the human body, including those in our skin, muscles, nerves, intestines, blood and bones.
3 to 5
Believe it or not, that’s the approximate number of pounds of bacteria you’re carrying around, depending on your size. Even though bacterial cells greatly outnumber ours, they’re much smaller than our cells and therefore account for less than 3 percent of our body mass. Scientists are learning more about how our body bacteria contribute to our health.
24
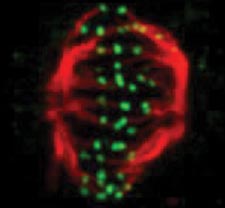
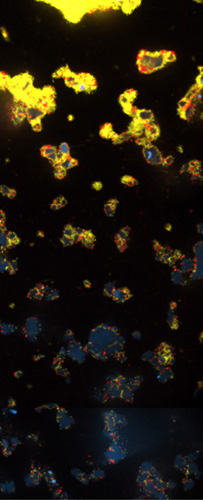
A snapshot of a phase of the cell cycle. Credit: Jean Cook and Ted Salmon, UNC School of Medicine.
This is the typical length in hours of the animal cell cycle, the time from a cell’s formation to when it splits in two to make more cells.
120
That’s the approximate lifespan in days of a human red blood cell. Other cell types have different lifespans, from a few weeks for some skin cells to as long as the life of the organism for healthy neurons.
50 to 70 billion
Each day, approximately this many cells die in the human body as part of a normal process that serves a healthy and protective role. Those that die in the largest numbers are skin cells, blood cells and some cells that line structures like organs and glands.