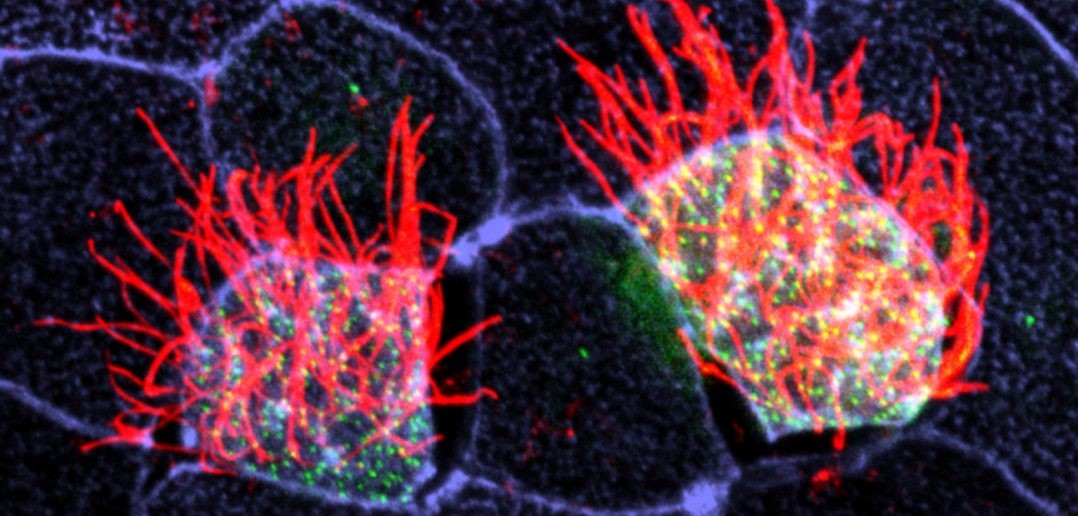
 Cells covered with cilia (red strands) on the surface of frog embryos. Credit: The Mitchell Lab.
Cells covered with cilia (red strands) on the surface of frog embryos. Credit: The Mitchell Lab.The outermost cells that line blood vessels, lungs, and other organs also act like guards, alert and ready to thwart pathogens, toxins, and other invaders that can do us harm. Called epithelial cells, they don’t just lie passively in place. Instead, they communicate with each other and organize their internal structures in a single direction, like a precisely drilled platoon of soldiers lining up together and facing the same way.
Lining up this way is crucial during early development, when tissues and organs are forming and settling into their final positions in the developing body. In fact, failure to line up in the correct way is linked to numerous birth defects. In the lungs, for instance, epithelial cells’ ability to synchronize with one another is important since these cells have special responsibilities such as carrying mucus up and out of lung tissue. When these cells can’t coordinate their functions, disease results.
Some lung epithelial cells are covered in many tiny, hair-like structures called cilia. All the cilia on lung epithelial cells must move uniformly in a tightly choreographed way to be effective in their mucus-clearing job. This is a unique example of a process called planar cell polarity (PCP) that occurs in cells throughout the body. Researchers are seeking to identify the signals cells use to implement PCP.
In one recent study, NIGMS grantee Brian Mitchell and his colleagues at Northwestern University’s Feinberg School of Medicine in Chicago, Ill, looked at a protein called CLAMP/Spef1 and its role in facilitating PCP signaling between cells. CLAMP/Spef1 molecules stick to microtubules, small structures that form the cell’s skeleton. These structures are important in helping the cell orient itself with other cells in PCP. In addition to forming on the microtubules, CLAMP/Spef1 can be found along the roots of cilia and within the membranes of cells near where they touch other cells. When Mitchell’s team depleted the normal amounts of CLAMP in frog embryo cells, the cells lost their PCP orientation, and their cilia did not coordinate and move in a single direction. Based on those findings, the researchers believe CLAMP is involved in cell-to-cell communication. Their next step will be to determine exactly how CLAMP aids in that communication.
Mitchell’s research is funded by NIGMS grant R01GM089970.

