This time of year, many creepy crawlies take center stage to frighten people of all ages. To celebrate Halloween, we’ve conjured up a slideshow of fascinating but spooky species that NIGMS-funded scientists study. Some of these creatures drink blood like vampires, while others—frogs, worms, flies, and salamanders—are perfect cauldron ingredients for a witch’s brew. Check out the slideshow—if you dare!
Continue reading “Slideshow: Creepy Crawlies”Tag: Cool Videos
Science Snippet: Zooming In on Nanoparticles
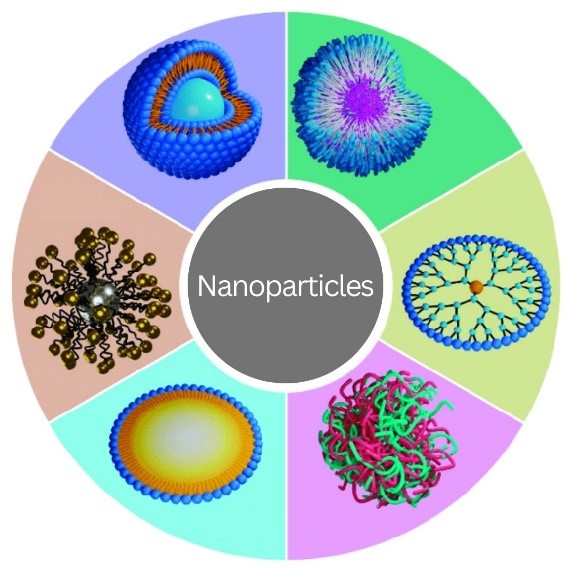
Nanoparticles may sound like gadgets from a science fiction movie, but they exist in real life. They’re particles of any material that are less than 100 nanometers (one-billionth of a meter) in all dimensions. Nanoparticles appear in nature, and humans have, mostly unknowingly, used them since ancient times. For example, hair dyeing in ancient Egypt involved lead sulfite nanoparticles, and artisans in the Middle Ages added gold and silver nanoparticles to stained-glass windows. Over the past several decades, researchers have studied nanoparticles for their potential uses in many fields, from computer engineering to biology.
A nanoparticle’s properties can differ significantly from those of larger pieces of the same material. Properties that may change include:
Continue reading “Science Snippet: Zooming In on Nanoparticles”Award-Winning Safety Training Videos Showcase Inclusivity in the Lab
Virginia Commonwealth University’s (VCU’s) Center on Health Disparities and safety and risk management department in Richmond teamed up to develop a series of six lab safety training videos with supplemental funding to their NIGMS-funded Initiative for Maximizing Student Development (IMSD) program. The videos cover topics such as safety culture, biosafety, chemical safety, and emergency response, but what sets them apart is how they showcase diversity and inclusion in the lab.
The first video in the safety training series describes the importance of maintaining positive safety culture, which includes people’s perceptions and attitudes toward safety. This video, along with the other five, is on our NIGMS laboratory safety training and guidelines webpage.
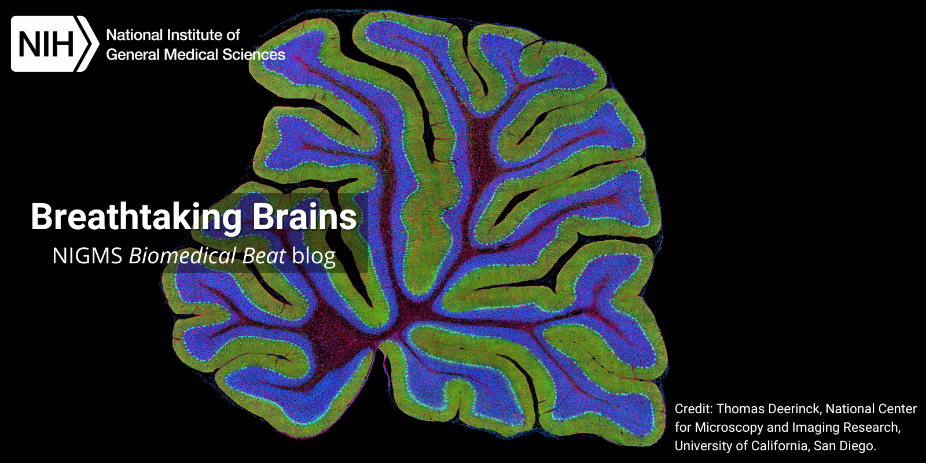
Slideshow: Breathtaking Brains
The average human brain is only about 3 pounds, but this complex organ punches well above its weight, acting as the control center for the whole body. Many of the brain’s intricacies still aren’t fully understood. To gain more insight into brain processes, scientists often peer into the brains of research organisms such as fruit flies and mice. These organisms have shed light on how our brains maintain circadian rhythms, how neuropsychiatric disorders develop, and more.
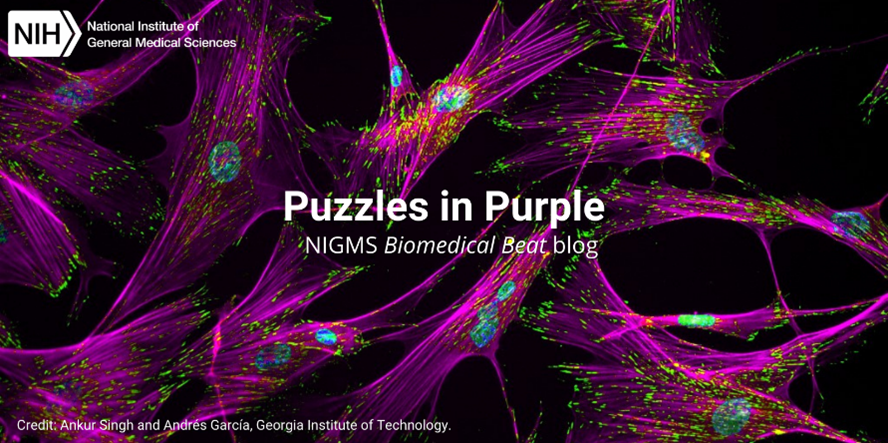
Continue reading “Slideshow: Breathtaking Brains”Photo Quiz: Puzzles in Purple
To make naturally colorless biological structures easier to study, scientists often use fluorescent tags and other tools to color them. Here, we feature images with purple hues and pair them with questions to test your knowledge of basic science concepts.
Visit our image and video gallery for more scientific photos, illustrations, and videos in all the colors of the rainbow.
Continue reading “Photo Quiz: Puzzles in Purple”Slideshow: Mitosis Masterpieces
The intricate process of mitosis—a cell splitting into two identical daughter cells—plays a pivotal role in sustaining life. Many scientists study this process to understand what’s needed for it to progress normally and why it sometimes goes awry, such as in cancer. During their research, the scientists often create eye-catching images and videos, and we showcase some of those visuals here.
Continue reading “Slideshow: Mitosis Masterpieces”Cool Video: A Biological Lava Lamp
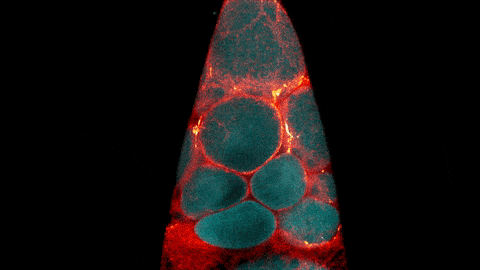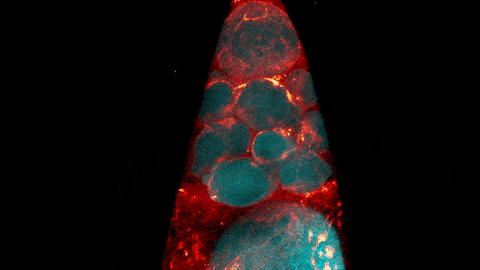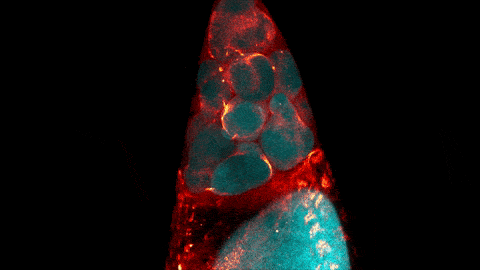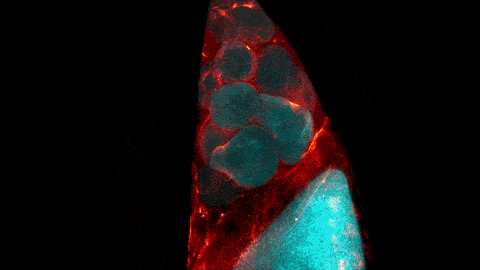
What looks like a bubbling lava lamp is actually part of an egg cell’s maturation process. In many animals, the egg cell develops alongside sister cells. These sister cells are called nurse cells in the fruit fly (Drosophila melanogaster), and their job is to “nurse” an immature egg cell, or oocyte. Toward the end of oocyte development, the nurse cells transfer all their contents into the oocyte in a process called nurse cell dumping. This video captures this transfer, showing significant shape changes on the part of the nurse cells (blue), which are powered by wavelike activity of the protein myosin (red).
Researchers created the video using a confocal laser scanning microscope. Learn about this type of microscope and other scientific imaging tools by stepping into our virtual imaging lab, and check out more basic science videos and photos in the NIGMS Image and Video Gallery.

This post is a great supplement to Pathways: The Imaging Issue.
The video was taken using a confocal laser scanning microscopy (sometimes shortened to just “confocal microscopy”), one of the techniques mentioned in the Pathways timeline (1970s).
Learn more in our Educator’s Corner.
Career Conversations: Q&A with Microbiologist Josephine Chandler
Josephine (Josie) Chandler, Ph.D., first became interested in science when she took a high school chemistry class. In college, she fell in love with microbiology and ultimately earned a Ph.D. in the field. Today, she’s an associate professor of molecular biosciences at the University of Kansas in Lawrence, where her lab investigates interactions in bacterial communities. By better understanding these interactions, scientists may find new ways to stop infections or break down environmental pollutants—a process known as bioremediation.
Continue reading “Career Conversations: Q&A with Microbiologist Josephine Chandler”Career Conversations: Q&A with Organic Chemist Osvaldo Gutierrez
Osvaldo Gutierrez, Ph.D., was born in Rancho Los Prietos, a small town in central Mexico where his grandmother served as a midwife. Seeing how his grandmother helped people through her work inspired Dr. Gutierrez to pursue a career where he, too, could help people. His family emigrated to the United States when he was young. Despite challenges he faced in a new country, he graduated from high school, attended community college, and was accepted to the University of California, Los Angeles. He originally planned to become a medical doctor, but an undergraduate research experience sparked an interest in chemistry, and he ultimately earned a Ph.D. in the field.
Continue reading “Career Conversations: Q&A with Organic Chemist Osvaldo Gutierrez”Expert Advice on Starting a Lab
During our Starting Your Own Lab webinar, attendees asked so many insightful questions that we ran out of time to respond to all of them. So we asked nine NIGMS early career investigators to tackle the most popular ones in short videos, which were featured on our social media. Now, you can watch the whole series on our YouTube channel.
1. What advice do you have for postdocs searching for a faculty position?Continue reading “Expert Advice on Starting a Lab”