Stem cells are a special type of cell that is uniquely able to replicate many times and to develop into many different cell types. Stem cells live in tissues and organs throughout the body—including the liver, muscles, and skin—at all stages of life.
Continue reading “What Are Stem Cells?”Tag: Stem Cells
From Fireflies to Physiology: Q&A With Yvon Woappi

“In high school, one of my teachers encouraged me to take an advanced biology class, and I’m so glad I did,” says Yvon Woappi, Ph.D. “The class opened my eyes to the fact that there were other people who loved nature like I did—they’re called biologists!” Now, Dr. Woappi is an assistant professor of physiology and cellular biophysics at Columbia University in New York City. We talked with him about his early love of nature and the night sky, the support he received from NIGMS training programs, and his research on wound healing.
Get to Know Dr. Woappi
- Coffee or tea? Tea
- Favorite music genre? Makossa, which originated in Douala, Cameroon
- Cats or dogs? Cats
- Rainy or sunny? Rainy
- Ocean or lake? Ocean
- Childhood dream job? Painter
- Favorite hobby? Chess
- Favorite lab tool? Fluorescent microscope
- Favorite pipette size? 100 microliters
- A scientist (past or present) you’d like to meet? Jonas Salk (who developed a safe and effective polio vaccine)
Surgeon Chris McCulloh Stands Up to Disability

Credit: Chris McCulloh.
Chris McCulloh
Job: 4th-year general surgical resident, Morristown Medical Center in New Jersey
Grew up in: Manhattan
When not at work, he’s: Programming, coding, thinking about artificial intelligence, and machine learning
Hobbies: Writing/producing electronic music, weightlifting
Ten years ago, Chris McCulloh planned to enter medical school and fulfill his dream of becoming a surgeon. Instead, just months before he was to start med school, he ended up a patient. A freak accident—slipping on a hardwood floor, flying backwards, and landing neck-first on the edge of a glass coffee table—left him with both legs paralyzed at age 28. Undaunted, he deferred entering medical school for a year, undergoing surgery and spending months in rehab.
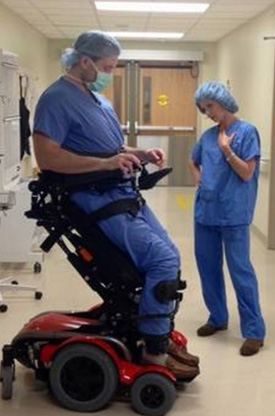
McCulloh prepares for surgery while “20/20” host Elizabeth Vargas stands alongside him as part of a 2017 interview.
Credit: Morristown Medical Center.
McCulloh has since finished medical school and recently completed a 2-year pediatric surgery research fellowship at Nationwide Children’s Hospital in Columbus, Ohio. He is now two-thirds of the way through his surgical residency at the Morristown (New Jersey) Medical Center, thanks to the assistance of a specialized wheelchair that allows him to stand nearly to his 6-foot-3 height and helps him perform five to six surgeries a day.
He’s received plenty of attention for being a surgeon with a disability. Along with several print media stories, he was interviewed in 2013 for CBS’ “The Doctors,” and in 2017, ABC’s “20/20” included McCulloh in an episode on physicians with disabilities. But it’s not the wheelchair that distinguishes McCulloh, says Gail Besner, a pediatric surgeon and researcher who hired McCulloh as a postdoctoral fellow. Rather, it’s his enthusiasm, natural research skills, and exceptional surgical prowess that make him special. Besner sees no reason why he won’t reach his goal of landing a highly competitive pediatric surgical residency. “I think he’s capable of doing anything he puts his mind to,” she says.
Continue reading “Surgeon Chris McCulloh Stands Up to Disability”
Stem Cells Do Geometry
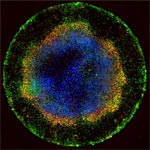
Each fluorescent point of light making up the multicolored rings in this image is an individual human embryonic cell in the early stages of development. Scientists seeking to understand the molecular cues responsible for early embryonic patterning found that human embryonic cells confined to areas of precisely controlled size and shape begin to specialize, migrate and organize into distinct layers just as they would under natural conditions.
Read the Inside Life Science article to learn more about this research, which has opened a new window for studying early development and could advance efforts aimed at using human stem cells to replace diseased cells and regenerate lost or injured body parts.
Cool Image: Of Surfaces and Stem Cells
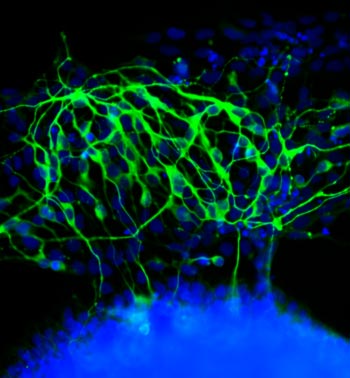
If you think this image looks like the fluorescent outline of a brain, you’re on the right track. The green threads show neurons that have just formed from unspecialized cells called stem cells.
Researchers led by Laura Kiessling ![]() of the University of Wisconsin-Madison directed the stem cells to become neurons by changing the quality of the surface on which they grew. In experiments testing different gels used to grow stem cells in the lab, the scientists found that the stiffness of those gels influenced cell fate decisions.
of the University of Wisconsin-Madison directed the stem cells to become neurons by changing the quality of the surface on which they grew. In experiments testing different gels used to grow stem cells in the lab, the scientists found that the stiffness of those gels influenced cell fate decisions.
When grown on a soft gel with a brain tissue-like surface, the stem cells began to transform into neurons. This happened without the addition of any of the proteins normally used to coax stem cells to specialize into different types of cells.
A better understanding of how stem cell fate is influenced by the mechanical properties of a surface could help researchers who are trying to harness the blank slate cells for tissue regeneration or other therapeutic uses.
This work also was funded by NIH’s National Institute of Biomedical Imaging and Bioengineering; National Heart, Lung, and Blood Institute; and National Institute of Neurological Disorders and Stroke.
Learn more:
University of Wisconsin-Madison News Release ![]()
Two Proteins That Regulate Energy Use Play Key Role in Stem Cell Development
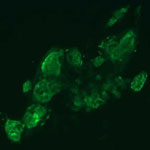
Hannele Ruohola-Baker and a team of researchers at the University of Washington recently discovered that two proteins responsible for regulating how cells break down glucose are also essential for stem cell development. The scientists showed that the proteins HIF1 alpha and HIF2 alpha are both required to reprogram adult human cells into pluripotent stem cells, which have the ability to mature into any cell type in the body. Taking a closer look at what each protein does on its own, the researchers found that HIF1 alpha was beneficial for reprogramming throughout the process, whereas HIF2 alpha was required at early stages but was detrimental at later stages of reprogramming. Because the two proteins also play a role in transforming normal cells into cancer cells, the findings could lead to future advances in cancer research.
Learn more:
University of Washington News Release
Ruohola-Baker Lab
Once Upon a Stem Cell Article from Inside Life Science
Learning About Cancer by Studying Stem Cells Article from Inside Life Science
Sticky Stem Cells Article from Inside Life Science
Healing Wounds, Growing Hair
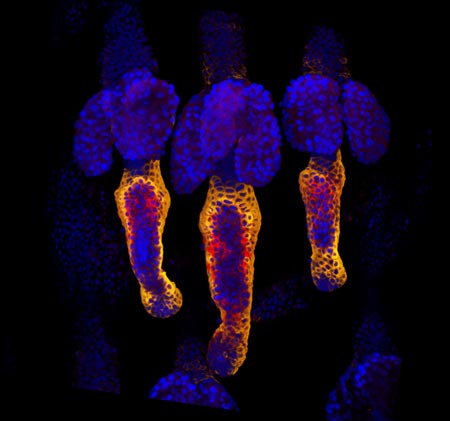
Whether injured by a scrape, minor burn or knife wound, skin goes through the same steps to heal itself. Regrowing hair over new skin is one of the final steps. All the hair you can see on your body is non-living, made up of “dead” cells and protein. It sprouts from living cells in the skin called hair follicle stem cells, shown here in red and orange. For more pictures of hair follicle stem cells—and many other stunning scientific images and videos—go to the NIGMS Image and Video Gallery.
Learn more:
Protein May Help Reduce Intestinal Injury
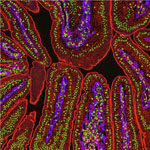
Gail Besner of Nationwide Children’s Hospital and her research team recently found out how the HB-EGF growth factor protein could potentially aid the development of treatments for a number of conditions. Using model systems in two separate studies, the scientists discovered that HB-EGF could protect the intestines from injury by stimulating cell growth and movement and by decreasing substances formed upon intestinal injury that worsen the damage. They also showed that administration of mesenchymal stem cells could further shield the intestines from injury. Future treatments involving a combination of HB-EGF and stem cells could, for example, help cancer patients sustain fewer intestinal injuries resulting from radiation therapy.
This work also was funded by NIH’s National Institute of Diabetes and Digestive and Kidney Diseases.
Learn more:
Nationwide Children’s Hospital News Release
Making Strides in Genomic Engineering of Human Stem Cells
Human pluripotent stem cells (hPSCs) can multiply indefinitely and give rise to virtually all human cell types. Manipulating the genomes of these cells in order to remove, replace or correct specific genes holds promise for basic biomedical research as well as medical applications. But precisely engineering the genomes of hPSCs is a challenge. A research team led by Erik Sontheimer of Northwestern University and James Thomson of the Morgridge Institute for Research at the University of Wisconsin-Madison developed a technique that could be a great improvement over existing, labor-intensive methods. Their approach uses an RNA-guided enzyme from Neisseria meningitidis bacteria—part of a recently discovered bacterial immune system—to efficiently target and modify specific DNA sequences in the genome of hPSCs. The technique could eventually enable the repair or replacement of diseased or injured cells in people with some types of cancer, Parkinson’s disease and other illnesses.
This work also was funded by NIH’s National Center for Advancing Translational Sciences.
Learn more:
Silencing Extra Copy of Chromosome 21
Each year about 1 in 700 babies is born with Down syndrome, a condition that occurs when cells contain three copies of chromosome 21. A new technique offers a proof of principle for silencing the extra copy. Using induced pluripotent stem cells derived from a person with Down syndrome, a research team led by Jeanne Lawrence of the University of Massachusetts Medical School inserted a gene called XIST into the extra chromosome 21. The gene, which normally turns off one whole X chromosome in females, rendered the chromosome copy and most of its genes inactive. The researchers plan to test the approach in a mouse model of Down syndrome and use it to further explore the biology of chromosome errors. The findings could eventually aid the development of therapies to mitigate resulting medical problems.
This work also was funded by NIH’s National Cancer Institute and Eunice Kennedy Shriver National Institute of Child Health and Human Development.
Learn more:
University of Massachusetts Medical School News Release
Lawrence Lab ![]()