The cytoskeleton is a collection of fibers that gives shape and support to cells, like the skeleton does for our bodies. It also allows movement within the cell and, in some cases, by the entire cell. Three different types of fibers make up the cytoskeleton: actin filaments, intermediate filaments, and microtubules.
Powering Muscles
Actin filaments contract or lengthen to give cells the flexibility to move and change shape. Along with the protein myosin, they’re responsible for muscle contraction, including voluntary movement and involuntary muscle contractions, such as our heartbeats. Actin filaments are the thinnest and most brittle of the cytoskeletal fibers, but they’re also the most versatile in terms of shape.
Continue reading “Science Snippet: Learn About the Cytoskeleton”

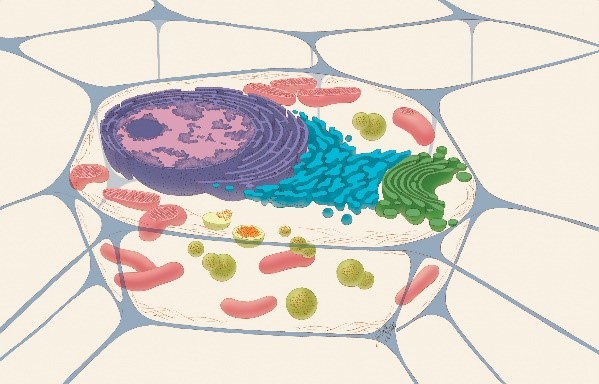 A cross-section of a cell showing organelles. Credit: Judith Stoffer.
A cross-section of a cell showing organelles. Credit: Judith Stoffer.
 Vern Schramm, professor of biochemistry at Albert Einstein College of Medicine, Bronx, New York. Credit: Albert Einstein College of Medicine.
Vern Schramm, professor of biochemistry at Albert Einstein College of Medicine, Bronx, New York. Credit: Albert Einstein College of Medicine.
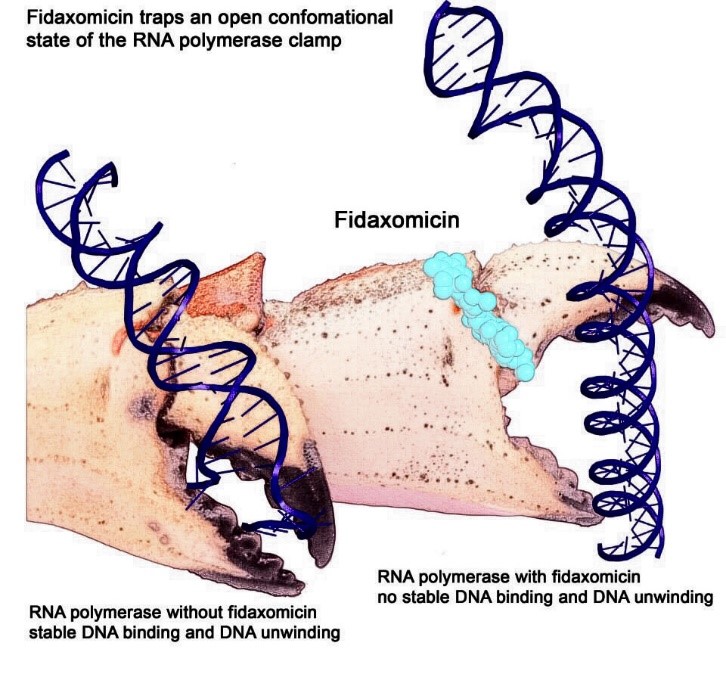 Artist interpretation of RNAP grasping and unwinding a DNA double helix. Credit: Wei Lin and Richard H. Ebright.
Artist interpretation of RNAP grasping and unwinding a DNA double helix. Credit: Wei Lin and Richard H. Ebright.