According to a recent estimate, implant infections following hip and knee replacement surgeries in the U.S. may number 65,000 by 2020, with the associated healthcare costs exceeding $1 billion. A new small, high-tech device could have a significant impact on improving health outcomes and reducing cost for these types of surgeries. The device, Air Barrier System (ABS), attaches on top of the surgical drape and gently emits HEPA-filtered air over the incision site. By creating a “cocoon” of clean air, the device prevents airborne particles—including the bacteria that can cause healthcare-associated infections—from entering the wound.
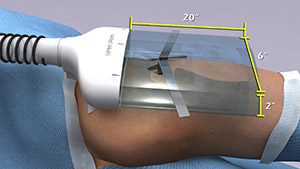
Scientists recently analyzed the effectiveness of the ABS device in a clinical study—funded by NIGMS—involving nearly 300 patients. Each patient needed an implant, such as an artificial hip, a blood vessel graft in the leg or a titanium plate in the spine. Because implant operations involve inserting foreign materials permanently into the body, they present an even higher risk of infection than many other surgeries, and implant infections can cause life-long problems.
The researchers focused on one of the most common causes of implant infections—the air in the operating room. Although operating rooms are much cleaner than almost any other non-hospital setting, it’s nearly impossible to sterilize the entire room. Instead, the scientists focused on reducing contaminants directly over the surgical site. They theorized that if the air around the wound was cleaner, the number of implant infections might go down.
Measuring the levels of airborne bacteria and particles was laborious. The researchers sampled air every 10 minutes during surgeries—nearly 3,000 times over a total of 470 hours.
The results proved that the ABS successfully cleansed air around incision sites—the average number of airborne particles dropped by 37 to 91 percent, depending on the type of surgery. The device also kept airborne microorganisms to almost zero. Most significant of all, the ABS appeared to reduce implant infections. In the group of patients whose surgeries used standard antiseptic techniques, there were four implant infections. In the group that added the ABS, there were none.
The ABS device is relatively inexpensive, costing $200-250 per surgery. Treating a single post-surgical infection can be far costlier—an estimated $40,000 for each of the four infections in this study. So using the device—which has been approved for sale by the U.S. Food and Drug Administration—promises not only to reduce the post-surgical implant infections, but might even reduce overall healthcare costs.


This is novel way to help reduce implant infection in ORs that should be embraced by the medical community. With such little cost and effort, ortho, vascular, and spine procedures can be made much safer. With the concentrated effort on reducing hospital acquired infections, surgeons and hospital administrators should seriously consider this technology.
These types of air purification using Hepa Filters to reducecontamination has been commonly if not routine in tissue culture and many biotechnology procedures and should have been either considered or evaluated years ago for surgical theaters.
Engineers have been doing this for over five decades in clean rooms for semiconductor integrated circuit production. Computer chip fabrication cannot tolerate any particles and all work areas are bathed in HEPA-filtered air. What took surgery so long?
I agree. Semiconductor manufacturing, tissue harvesting, pharmaceutical and sterile medical devices have robust and are frequently monitored and validated for cleanliness. There are a couple studies that show the OR door opens an average of 6 times per 10 minutes in joint replacement procedures. Think about that… that is 40-60 times during an implant procedure.
What are the advantages and disadvantages of using this method?
The advantage is that the device will greatly reduce the chance of infection during surgery (but it won’t eliminate the risk entirely). The disadvantage is that using the device makes the surgery slightly more expensive (an additional $200 to $250 per surgery).
Yes, I agree. Semiconductor manufacturing, tissue harvesting, pharmaceutical and sterile medical devices have robust and are frequently monitored and validated for cleanliness. There are a couple studies that show the OR door opens an average of 6 times per 10 minutes in joint replacement procedures. Thanks for sharing the great information with us.