Of the 118 known elements, scientists believe that 25 are essential for human biology. Four of these (hydrogen, oxygen, nitrogen, and carbon) make up a whopping 96 percent of our bodies. The other 21 elements, though needed in smaller quantities, perform fascinating and vital functions. Phosphorus is one such element. It has diverse uses outside of biology. For example, it can fuel festive Fourth of July fireworks! Inside our bodies, it’s crucial for a wide range of cell functions.
 Phosphorus plays a vital role in life as part of DNA’s backbone. Red phosphorus helps ignite matches, and white phosphorus glows in the presence of oxygen. Credit: Compound Interest.
Phosphorus plays a vital role in life as part of DNA’s backbone. Red phosphorus helps ignite matches, and white phosphorus glows in the presence of oxygen. Credit: Compound Interest. CC BY-NC-ND 4.0
 . Click to enlarge
. Click to enlarge
A Volatile and Versatile Discovery
 Joseph Wright of Derby’s The Alchemist Discovering Phosphorus depicts Hennig Brand’s discovery of phosphorus.
Joseph Wright of Derby’s The Alchemist Discovering Phosphorus depicts Hennig Brand’s discovery of phosphorus.
Phosphorus is the first element with a recorded discovery story. Today, we extract it from phosphate rocks, but it was first isolated from human urine in 1669. In an attempt to create the philosopher’s stone (a substance, according to legend, that could turn metals into gold), German alchemist Hennig Brand boiled down a large amount of human urine into a paste. He then heated the paste until it produced a vapor that condensed into white, waxy droplets. Because the new substance glowed, he named it “phosphorus,” from the Greek word for “light-bearer.”
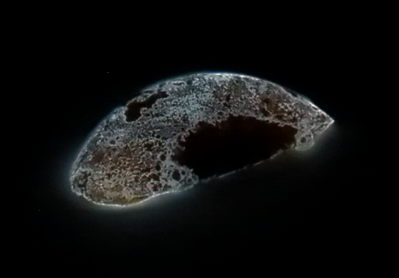 White phosphorus glows in the dark. Credit: Endimion17. CC-BY-SA 3.0
White phosphorus glows in the dark. Credit: Endimion17. CC-BY-SA 3.0  .
.
Phosphorus comes in two main forms. White phosphorus, the type that Brand collected, is poisonous, can cause severe burns, and spontaneously bursts into flame at around 86 degrees Fahrenheit if exposed to air. Its volatility makes it useful for flares, fireworks, and weaponry. Scientists later discovered the other main form of phosphorus, red phosphorus, which is stable, nontoxic, and doesn’t glow. You can find red phosphorus on the striking surface of safety match boxes.
These uses, along with applications in processes such as steel manufacturing and fine china production, make up a small percentage of the uses for phosphorus in the United States. The vast majority of U.S. phosphorus is used to produce fertilizers and supplements for livestock because phosphorus is an essential mineral for plants and animals.
The “Us” in Phosphorus
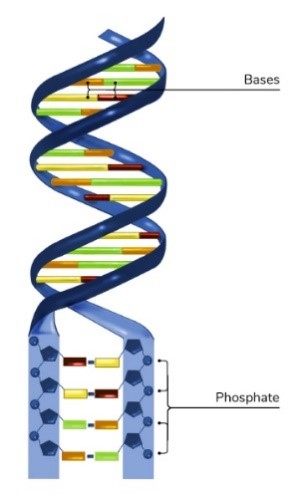 Phosphorus helps hold the DNA double helix together. Credit: NIGMS.
Phosphorus helps hold the DNA double helix together. Credit: NIGMS.
Like other living things, we need phosphorus in our diets, and getting enough of the mineral from our food is easy. Phosphorus deficiencies are very rare and are usually only seen when people are near starvation.
On average, our bodies contain about 750 grams of phosphorus, and about 85 percent of it is stored in our bones and teeth. But the phosphorus in our bodies isn’t found on its own. Instead, it’s found in compounds with other molecules. In addition to strengthening our bones, these compounds are important components of cell membranes and of our cells’ energy currency, adenosine triphosphate (ATP). Phosphorus-containing compounds also activate many enzymes and hormones, as well as help red blood cells deliver oxygen to different parts of our bodies.
Even our DNA needs phosphorus. DNA molecules look like twisted ladders; they have two long sides with rungs, called bases, in between. The bases carry hereditary information, and the long sides keep the bases organized. Phosphate, a compound made up of a phosphorus atom and four oxygen atoms, connects the sugars that make up the long sides, or backbones, of DNA.
Overall, for the small percentage of our bodies that phosphorus makes up, it has a huge impact on our biology. It doesn’t have the power of the philosopher’s stone that Brand was searching for when he discovered the element, but it’s even more valuable.
NIGMS Research Support
NIGMS funds a wide range of research focused on improving the molecular-level understanding of basic biological processes. These studies include exploring phosphorus’ role in such processes. Ultimately, understanding the function of elements and molecules in biological systems can help researchers develop ways to detect or treat diseases.
Check out our other posts on elements.


This is an excellent little summary of the history and usefulness of phosphorous, and a refresher on its place in chemistry and biology.
Nice and brief.
Interesting and very concise!