Most of the mouthwatering dishes in a Thanksgiving feast share a vital ingredient: salt! Though the words “salt” and “sodium” are often used interchangeably, table salt is actually a compound combining the elements sodium and chloride. Table salt is the most common form that sodium takes on Earth. Many other sodium compounds are also useful to us. For instance, you might use baking soda, also known as sodium bicarbonate, in preparing Thanksgiving treats. Sodium compounds are also used in soaps and cosmetics and in producing paper, glass, metals, medicines, and more.
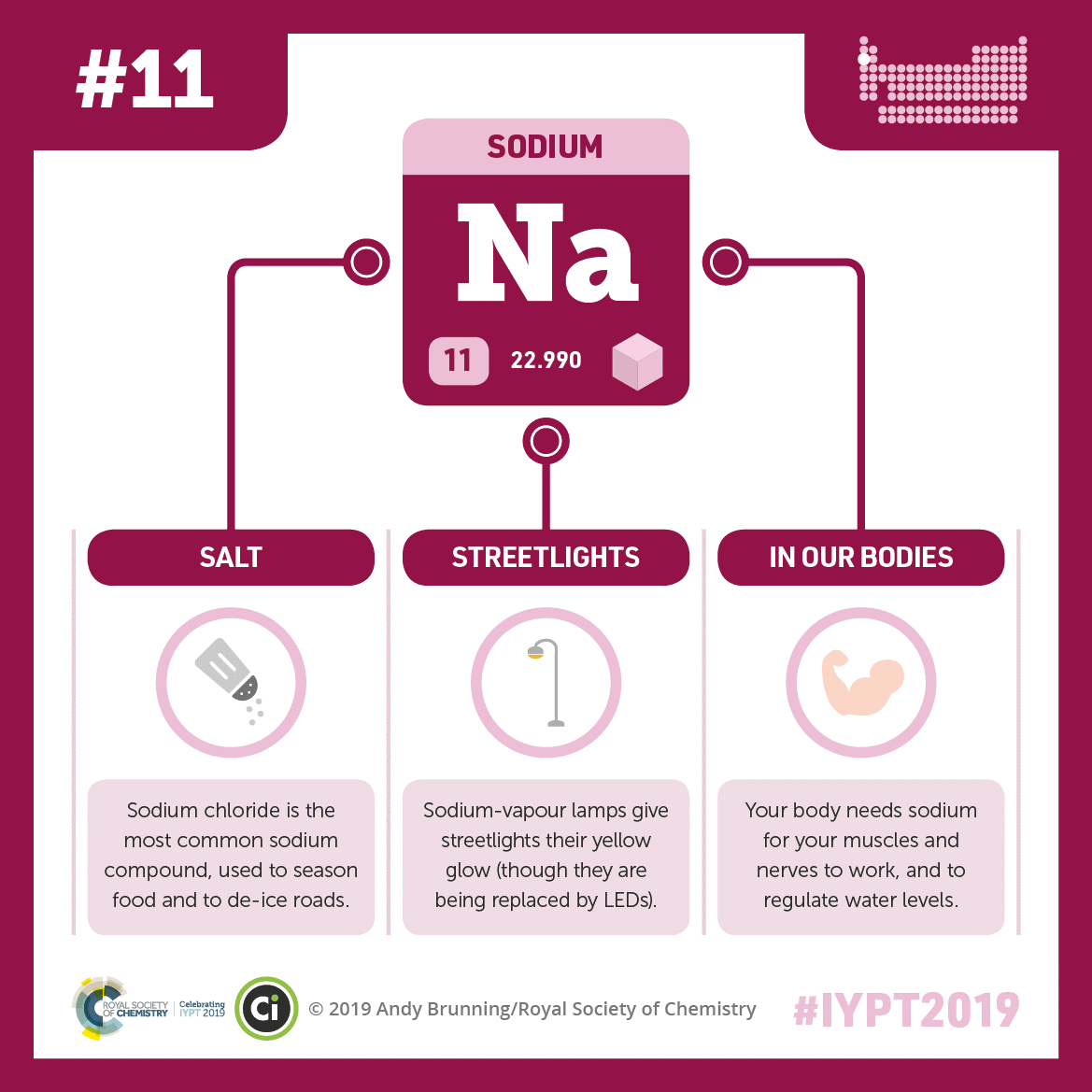 The best-known sodium compound is table salt (sodium chloride). Sodium also gives traditional streetlights their yellow glow and is essential for muscle and nerve function. Credit: Compound Interest. CC BY-NC-ND 4.0. Click to enlarge
The best-known sodium compound is table salt (sodium chloride). Sodium also gives traditional streetlights their yellow glow and is essential for muscle and nerve function. Credit: Compound Interest. CC BY-NC-ND 4.0. Click to enlarge
Cell Signaling
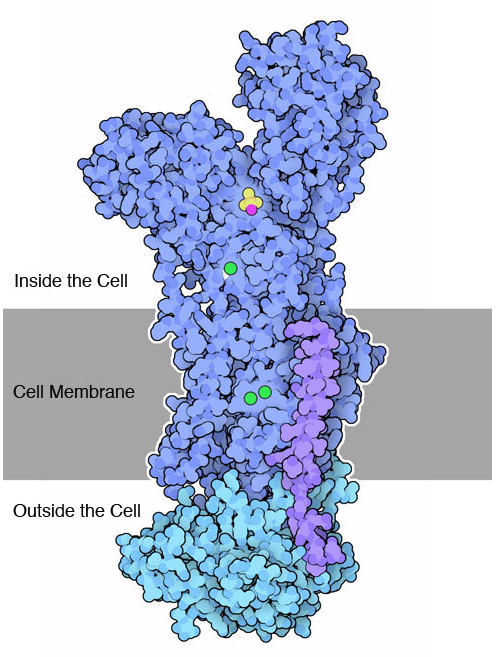 A sodium-potassium pump with potassium ions (green) moving through it. Credit: Protein Data Bank.
Click to enlarge
A sodium-potassium pump with potassium ions (green) moving through it. Credit: Protein Data Bank.
Click to enlarge
Sodium plays a vital role in our bodies. In partnership with another element, potassium, it acts like a chemical battery that powers our nerve impulses and muscle contractions. This battery is maintained by proteins called sodium-potassium pumps, which are embedded like tunnels in our cell membranes. These pumps move three sodium ions out of the cell while moving two potassium ions into it, leading to a significant difference in the concentrations of the two elements. When one nerve cell needs to communicate with another, it opens special channels that allow sodium to flood in. This rush of sodium tells the nerve cell to fire. The result is a chain reaction: The signal is carried from nerve cell to nerve cell until it reaches the brain or a muscle for action. When triggered by nerve signals, muscle cells alter their sodium/potassium balance as part of the process that they use to contract and move our bodies. The sodium-potassium balance is so important that sodium-potassium pumps use up about a third of our cell’s main energy source, a molecule called adenosine triphosphate.
Under Pressure
 Too much sodium can damage blood vessels and result in high blood pressure.
Too much sodium can damage blood vessels and result in high blood pressure.
Our bodies need a certain amount of sodium to help maintain a healthy balance of water and minerals. But if we consume too much sodium, it causes our bodies to hold on to more water than we need. This extra water increases the volume of blood in the bloodstream, which puts more pressure on blood vessels and forces the heart to work harder. Over time, the strain can stiffen blood vessels and lead to high blood pressure.
The majority of people in the United States consume more sodium than recommended, mostly from processed and restaurant foods. According to the 2015–2020 Dietary Guidelines for Americans, a healthy amount of sodium for adults is less than 2,300 milligrams—about a teaspoon—per day. Being mindful of sodium intake can help prevent high blood pressure and protect our health.
NIGMS-Supported Sodium Studies
NIGMS funds researchers who are investigating a range of topics related to sodium. Some of these scientists are:
- Developing tools to better understand the channels that enable sodium to flood into nerve and muscle cells.
- Investigating a signaling pathway that triggers sodium channels. The pathway could be a target for medications to treat pain, seizures, muscle spasms, or irregular heartbeats.
- Determining how sodium channels are affected by toxins from tarantula spiders, as a starting point for developing medications that target these channels.
Check out our other posts on elements.


Nice refresher and concise.
The article gives a good explanation of the role of sodium in our bodies.
By the way, could you describe the mechanism (process) by which the sodium (ion) makes the body to hold on to water which in turn increases the blood volume and consequently increases the blood pressure? Thanks!
Thanks for your question, Raj. When your body detects high amounts of sodium in your blood, it will pull water into the bloodstream to lower the salt concentration. Think of it as watering down a drink that’s too sweet or a soup that’s too salty–you’re adding water to adjust the concentration rather than removing some of the sugar or salt. That increased blood volume leads to more force pushing on your blood vessels, or an increase in blood pressure.
Very little is said about K—its functions, correct daily requirement, and sources. I understand that many people do not consume enough K. For well over 30-years I have used KCl rather than NaCl. As a pharmacist back in the 70s we sold a lot of it—“No Salt”. Please tell us about K, including its toxicity and how that can occur. Thanks
Roy, thanks for your comment and suggestion to consider a post discussing potassium in the future.