We couldn’t survive without proteins. They’re essential molecules that provide cells with structure, aid in chemical reactions, support communication, and much more. Portion out protein numbers with us below!
10 Trillion
That’s how many proteins scientists estimate are in each human cell.
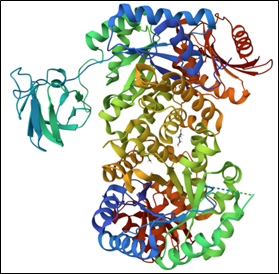
229,378
That’s how many structures researchers shared with the scientific community through the Protein Data Bank (PDB) from its establishment in 1971 to the end of 2024. The PDB is a global repository for 3D structural data of proteins, DNA, RNA, and even complexes these biological molecules form with medicines or other small molecules.
42
That’s the percent of your body weight (not counting water) that’s made up of proteins.
Continue reading “Proteins by the Numbers”