The tiny roundworm Caenorhabditis elegans is one of the most common research organisms—creatures scientists use to study life. While C. elegans may seem drastically different from humans, it shares many genes and molecular pathways with us. Viewed with a microscope, the worm can also be surprisingly beautiful. Aside from the stunning imagery, these examples from our Image and Video Gallery show how C. elegans helps scientists advance our understanding of living systems and find new ways to improve our health.
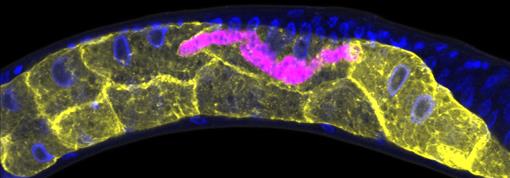 Credit: Keir Balla and Emily Troemel, University of California San Diego.
Credit: Keir Balla and Emily Troemel, University of California San Diego.
This C. elegans has been infected with microsporidia (purple), parasites closely related to fungi. The yellow shapes are the worm’s gut cells, and the blue dots are nuclei. Some microsporidia can infect people, so studying the parasites in worms could help researchers devise strategies to prevent or treat infections.
Continue reading “Cool Images: Wondrous Worms”

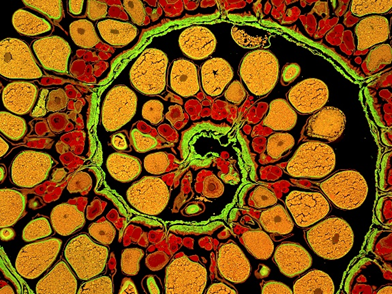 Credit: James E. Hayden, The Wistar Institute, Philadelphia, PA.
Credit: James E. Hayden, The Wistar Institute, Philadelphia, PA.
 A family of common marmosets. Credit: Francesco Veronesi.
A family of common marmosets. Credit: Francesco Veronesi.  Hawaiian bobtail squid. Credit: Dr. Satoshi Shibata.
Hawaiian bobtail squid. Credit: Dr. Satoshi Shibata.
 Viravuth (“Voot”) Yin, associate professor of regenerative biology and medicine at MDI Biological Laboratory and chief scientific officer at Novo Biosciences, Inc., in Bar Harbor, Maine. Credit: MDI Biological Laboratory.
Viravuth (“Voot”) Yin, associate professor of regenerative biology and medicine at MDI Biological Laboratory and chief scientific officer at Novo Biosciences, Inc., in Bar Harbor, Maine. Credit: MDI Biological Laboratory.
 Dr. Melissa Wilson.
Dr. Melissa Wilson.