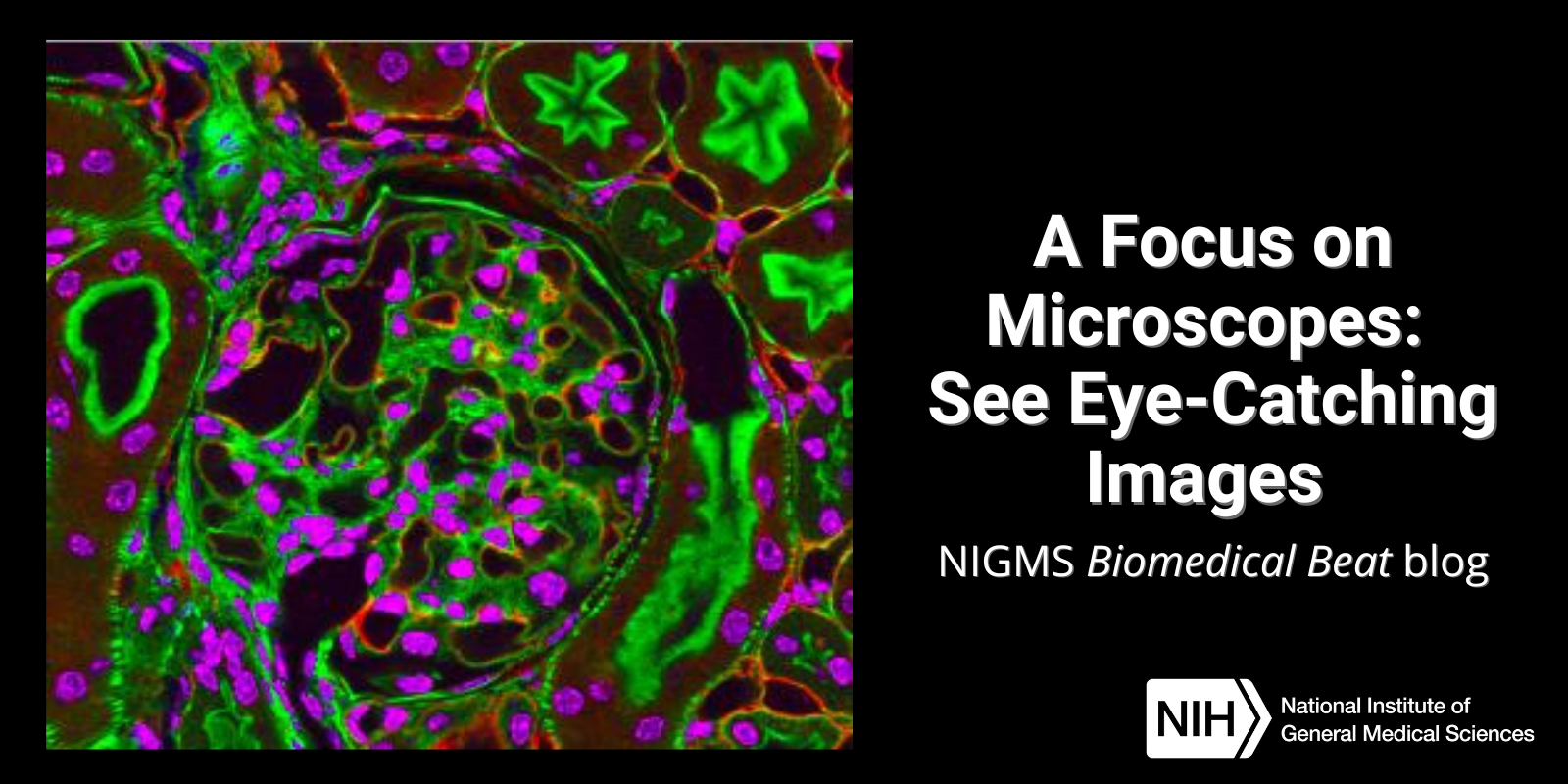
Have you ever wondered what creates striking images of cells and other tiny structures? Most often, the answer is microscopes. Many of us have encountered basic light microscopes in science classes, but those are just one of many types that scientists use. Check out the slideshow to see images researchers have captured using different kinds of microscopes. For even more images of the microscopic world, visit the NIGMS Image and Video Gallery.
Continue reading “A Focus on Microscopes: See Eye-Catching Images”Tag: Cool Tools/Techniques
Explore Scientific Imaging Through a Virtual “Internship”
Students, teachers, and other curious minds can step into a scientific imaging lab with a free online interactive developed by NIGMS and Scholastic. Imaging tools help scientists unlock the mysteries of our cells and molecules. A better understanding of this tiny world can help researchers learn about the body’s normal and abnormal processes and lead to more effective, targeted treatments for illnesses.
Pathways: The Imaging Issue
 Cover of Pathways student magazine.
Cover of Pathways student magazine.
NIGMS and Scholastic bring you our latest issue of Pathways, which focuses on imaging tools that help scientists unlock the mysteries of our cells and molecules. A better understanding of this tiny world can help researchers learn about the body’s normal and abnormal processes and lead to more effective, targeted treatments for illnesses.
Pathways is designed for students in grades 6 through 12. This collection of free resources teaches students about basic science and its importance to health, as well as exciting research careers.
Continue reading “Pathways: The Imaging Issue”Year in Review: Our Top Three Posts of 2020
Over the year, we dove into the inner workings of cells, interviewed award-winning researchers supported by NIGMS, shared a cool collection of science-themed backgrounds for video calls, and more. Here, we highlight three of the most popular posts from 2020. Tell us which of this year’s posts you liked best in the comments section below!
The Science of Infectious Disease Modeling
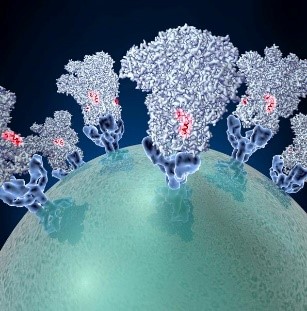 Spike proteins on the surface of a coronavirus. Credit: David Veesler, University of Washington.
Spike proteins on the surface of a coronavirus. Credit: David Veesler, University of Washington.
What does “modeling the spread” (or “flattening the curve”) mean, and how does it apply to infectious diseases such as COVID-19? Learn about the science of infectious disease modeling and how NIGMS supports scientists in the field.
Continue reading “Year in Review: Our Top Three Posts of 2020”An Enlightening Protein
 A fruit fly expressing GFP. Credit: Jay Hirsh, University of Virginia.
A fruit fly expressing GFP. Credit: Jay Hirsh, University of Virginia.
During the holiday season, twinkling lights are a common sight. But no matter what time of the year, you can see colorful glows in many biology labs. Scientists have enabled many organisms to light up in the dark—from cells to fruit flies and Mexican salamanders. These glowing organisms help researchers better understand basic cell processes because their DNA has been edited to express molecules such as green fluorescent protein.
Continue reading “An Enlightening Protein”Q&A With Nobel Laureate and CRISPR Scientist Jennifer Doudna
 Jennifer Doudna, Ph.D. Credit: University of California, Berkeley.
Jennifer Doudna, Ph.D. Credit: University of California, Berkeley.
The 2020 Nobel Prize in Chemistry was awarded to Jennifer Doudna, Ph.D., and Emmanuelle Charpentier, Ph.D., for the development of the gene-editing tool CRISPR. Dr. Doudna shared her thoughts on the award and answered questions about CRISPR in a live chat with NIH Director Francis S. Collins, M.D., Ph.D. Here are a few highlights from the interview.
Q: How did you find out that you won the Nobel Prize?
A: It’s a little bit of an embarrassing story. I slept through a very important phone call and finally woke up when a reporter called me. I was just coming out of a deep sleep, and the reporter was asking, “What do you think about the Nobel?” And I said, “I don’t know anything about it. Who won it?” I thought they were asking for comments on somebody else who won it. And she said, “Oh my gosh! You don’t know! You won it!”
Continue reading “Q&A With Nobel Laureate and CRISPR Scientist Jennifer Doudna”Freezing a Moment in Time: Snapshots of Cryo-EM Research
To get a look at cell components that are too small to see with a normal light microscope, scientists often use cryo-electron microscopy (cryo-EM). As the prefix cryo- means “cold” or “freezing,” cryo-EM involves rapidly freezing a cell, virus, molecular complex, or other structure to prevent water molecules from forming crystals. This preserves the sample in its natural state and keeps it still so that it can be imaged with an electron microscope, which uses beams of electrons instead of light. Some electrons are scattered by the sample, while others pass through it and through magnetic lenses to land on a detector and form an image.
Typically, samples contain many copies of the object a scientist wants to study, frozen in a range of orientations. Researchers take images of these various positions and combine them into a detailed 3D model of the structure. Electron microscopes allow us to see much smaller structures than light microscopes do because the wavelengths of electrons are much shorter than the wavelength of light. NIGMS-funded researchers are using cryo-EM to investigate a range of scientific questions.
Caught in Translation
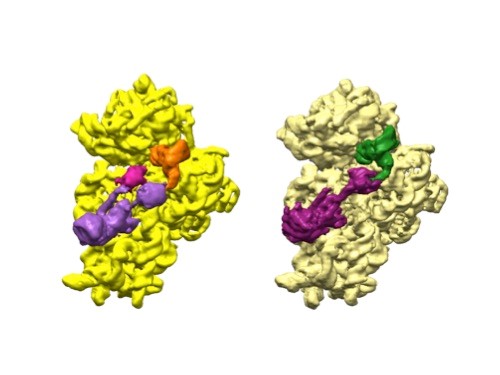 3D reconstructions of two stages in the assembly of the bacterial ribosome created from time-resolved cryo-EM images. Credit: Joachim Frank, Columbia University.
3D reconstructions of two stages in the assembly of the bacterial ribosome created from time-resolved cryo-EM images. Credit: Joachim Frank, Columbia University.
Joachim Frank, Ph.D., a professor of biochemistry and molecular biophysics and of biological sciences at Columbia University in New York, New York, along with two other researchers, won the 2017 Nobel Prize in Chemistry for developing cryo.
Dr. Frank’s lab focuses on the process of translation, where structures called ribosomes turn genetic instructions into proteins, which are needed for many chemical reactions that support life. Recently, Dr. Frank has adopted and further developed a technique called time-resolved cryo-EM. This method captures images of short-lived states in translation that disappear too quickly (after less than a second) for standard cryo-EM to capture. The ability to fully visualize translation could help researchers identify errors in the process that lead to disease and also to develop treatments.
Continue reading “Freezing a Moment in Time: Snapshots of Cryo-EM Research”Exploring Nature’s Treasure Trove of Helpful Compounds
 A cone snail shell. Credit: Kerry Matz, University of Utah.
A cone snail shell. Credit: Kerry Matz, University of Utah.
Over the years, scientists have discovered many compounds in nature that have led to the development of medications. For instance, the molecular structure for aspirin came from willow tree bark, and penicillin was found in a type of mold. And uses of natural products aren’t limited to medicine cabinet staples and antibiotics. A cancer drug was originally found in the bark of the Pacific yew tree, and a medication for chronic pain relief was first isolated from cone snail venom. Today, NIGMS supports scientists in the earliest stages of investigating natural products made by plants, fungi, bacteria, and animals. The results could inform future research and bring advances to the field of medicine.
Continue reading “Exploring Nature’s Treasure Trove of Helpful Compounds”Helium: An Abundant History and a Shortage Threatening Scientific Tools
Most of us know helium as the gas that makes balloons float, but the second element on the periodic table does much more than that. Helium pressurizes the fuel tanks in rockets, helps test space suits for leaks, and is important in producing components of electronic devices. Magnetic resonance imaging (MRI) machines that take images of our internal organs can’t function without helium. And neither can nuclear magnetic resonance (NMR) spectrometers that researchers use to determine the structures of proteins—information that’s important in the development of medications and other uses.
 Helium’s many uses include helping deep sea divers breathe underwater, airbags in cars to inflate, and magnets in MRI scanners to work properly. Credit: Compound Interest.
Helium’s many uses include helping deep sea divers breathe underwater, airbags in cars to inflate, and magnets in MRI scanners to work properly. Credit: Compound Interest.CC BY-NC-ND 4.0. Click to enlarge
Scientist Interview: Exploring the Promise of RNA Switches with Christina Dawn Smolke
Whether animals are looking for food or mates, or avoiding pathogens and predators, they rely on biosensors—molecules that allow them to sense and respond to their environments. Christina Dawn Smolke, Ph.D., a professor of bioengineering at Stanford University in California, focuses her research on creating new kinds of biosensors to receive, process, and transmit molecular information. Her lab has built RNA molecules, or switches, that can alter gene expression based on biochemical changes they detect.
Continue reading “Scientist Interview: Exploring the Promise of RNA Switches with Christina Dawn Smolke”